Kaiser Permanente researcher and colleagues reports new findings from long-term study in PLOS Medicine
Ten years ago, Kaiser Permanente Division of Research research scientist Erica P. Gunderson, PhD, MS, MPH, became the Principal Investigator on one of the world’s largest and most diverse prospective studies to look at future health outcomes in women who developed gestational diabetes (diabetes diagnosed for the first time during pregnancy) .

New findings from the Study of Women, Infant Feeding and Type 2 Diabetes After GDM Pregnancy (SWIFT) were published on May 20 in PLOS Medicine. The new research, Amino acid and lipid metabolism in post-gestational diabetes and progression to type 2 diabetes: A metabolic profiling study, conducted by Gunderson and her long-time collaborators at the University of Toronto, exemplifies what can be learned from a transdisciplinary approach that merges biochemistry and clinical epidemiologic research. The findings have also brought Gunderson a step closer to achieving one of the SWIFT study’s primary goals: developing a new blood test for early prediction of women with gestational diabetes who are at greatest risk of developing type 2 diabetes in the future. Gunderson talked about reaching the 10-year anniversary of the SWIFT study, the most recent findings, and where her research on maternal health is headed
Q: What makes the SWIFT study unique?
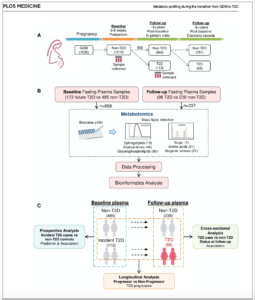
Gunderson: What makes the SWIFT study different from many other Kaiser Permanente research studies is that it doesn’t rely solely on electronic medical records. Our study team recruited 1,033 women with gestational diabetes during pregnancy for the SWIFT study and conducted research oral glucose tolerance tests and assessments from 2 months after delivery and beyond to build a biobank of blood samples and research databases. The SWIFT participants are an ethnically and racially diverse group of women who were diagnosed with gestational diabetes during a pregnancy between 2008 and 2011. Each woman consented to 3 in-person research visits, starting 6 to 9 weeks after giving birth and up to two years later, where we measured body weight, height, and waist circumference, and asked questions about breastfeeding and other lifestyle factors during the first year after delivery. At each study visit, each woman was also given a research oral glucose tolerance test. As a result, the blood samples we have collected over the past 10 years provide a rare resource to measure biomarkers preceding the development of type 2 diabetes in the SWIFT study participants.
 Q: What were the main findings from your new PLOS Medicine study?
Q: What were the main findings from your new PLOS Medicine study?
Gunderson: Right now, a 2-hour oral glucose tolerance test that requires a woman to fast for 8-14 hours is used to test for diabetes after pregnancy. Our goal for the SWIFT 10-year followup study is to develop an alternative blood test. The new study published in PLOS Medicine added more metabolites to our previous analysis. This helped identify an expanded group of metabolites that can predict with over 85% accuracy which women with gestational diabetes are at highest risk of progressing to type 2 diabetes. A blood test that looks at levels of these metabolites would allow obstetricians and primary care providers to identify the women most at risk for type 2 diabetes and advise them about changes in behavior to prevent future diabetes, such as breastfeeding, getting enough sleep, and healthful eating habits to avoid weight gain.
Q: What have you learned from following these women for 10 years?
Gunderson: There are other studies that have looked at progression to diabetes after pregnancy among women with gestational diabetes. But our study is the largest prospective study of women with gestational diabetes in which all participants were given standardized oral glucose tolerance tests from early postpartum and then tested for diabetes annually for up to 2 years. We’ve now seen about 20% of these women have developed diabetes within 8 to 10 years after pregnancy. In the general population, among women of this age, about 2% would would have progressed to diabetes within 10 years of giving birth. That means for the women with gestational diabetes, their risk is about 7 to 10-fold higher. My previously published studies have shown that women who breastfed for 5 to 6 months or more had half the risk of developing type 2 diabetes after gestational diabetes, independent of other factors, and that their infants had less rapid weight gain growth. Our ongoing studies will also provide insights into how the severity of gestational diabetes may affect a woman’s future risk for cardiovascular risk factors and outcomes.
Q: What will happen next?
Gunderson: Our research staff was ready to begin contacting the women in the SWIFT study for their 10-year follow-up visits to administer a fourth research blood test for type 2 diabetes when the COVID-19 pandemic hit. We will pick up where we left off, as soon as we can. The information we glean from the 10-year study research visits will bring us even closer to our goal of developing this test. It will also help us to identify metabolic differences that are associated with race and ethnicity that this test will need to take into account. Many thanks to all of the women who have stayed in the SWIFT research study for 10 years. Their decision to be part of this study and their continued participation will help many women in the future.
Read more about the study in this University of Toronto news story.
Funding for the new study in PLOS Medicine was supported by research grants from The National Institute of Child Health and Human Development (Gunderson, PI); The National Institute of Digestive, Diabetes and Kidney Disease (Gunderson, PI); Canadian Institutes of Health Research; and Janssen Pharmaceuticals Companies.
Coauthors of the new PLOS Medicine study include Mi Lai, Ying Liu, Anne Wu, Brian J. Cox, Feihan F. Dai, Hannes L. Röst, and Michael Wheeler of the University of Toronto and Gabriele V. Ronnett of Janssen Research & Development.




This Post Has One Comment
This is great! I can’t believe it has been 10 years already. Dr. Gunderson and the rest of the SWIFT STUDY team are amazing people to work with. I hope to work with you all again in the future.