Kaiser Permanente Vaccine Study Center begins inoculations as part of nationwide evaluation of Pfizer/BioNTech vaccine
About 75 children are scheduled to receive their first vaccination against COVID-19 as part of the Pfizer/BioNTech vaccine clinical trial through Kaiser Permanente Northern California.
Sacramento resident Luci Guardino, 11, was among the first children aged 5 to 11 to get the Pfizer vaccine. “I want other kids and people to know the COVID vaccine is safe and it will get us more back to a completely normal life…to do simple things like going to the movies or eating indoors or having friends over,” she said.
Guardino joins about 4,600 children at dozens of sites around the country participating in the Pfizer pediatric vaccine trial. The trial has a double-blind design so neither participants nor researchers know if a vaccine or placebo is being administered; two-thirds of the children will get the vaccine and one-third the placebo.

The children are getting a smaller dose of the vaccine than that given to adults and teens, though the rest of the vaccination process is the same: 2 doses 21 days apart, and careful monitoring for side effects or negative reactions. The trial continues to track participants for 2 years even if the vaccine becomes available for children before the trial ends.
Pfizer officials have suggested they may seek federal emergency use authorization to distribute the vaccine to children as early as September.
Guardino’s parents are both physicians with The Permanente Medical Group and support the trial’s move into younger patients. “Being a part of a large, important and historic study just seemed like a great opportunity and I was really proud of [our kids for] wanting to participate and help get closer back to normal,” said Luci’s mother, Stephanie Yee-Guardino, DO, a pediatrician at Kaiser Permanente South Sacramento.
She noted how much safer she felt after being vaccinated herself. “Getting our kids vaccinated and allowing them to experience that same reassurance when they are going to school, going to camps, doing things with their friends, doing sports, visiting family, is a wonderful thing.”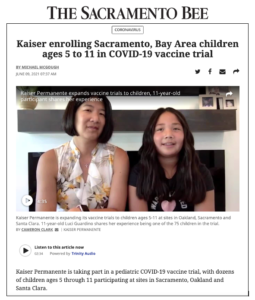
The Kaiser Permanente participation in the Pfizer pediatric trial is managed by the Kaiser Permanente Vaccine Study Center, based in Oakland and led by center Director Nicola Klein, MD, PhD. Klein emphasized the importance of vaccinating children even if they don’t get as sick from COVID-19 as older people do.
“Children and adolescents may benefit from having protection from COVID-19 so they can return to school, sports, and other activities safely,” said Klein, who is a research scientist with the Kaiser Permanente Division of Research. “Also, new and future variants raise the possibility that COVID-19 could behave differently in young people in the future, becoming more dangerous.”
She also noted that given the hospitalizations and deaths of children from COVID-19 so far, it qualifies as a serious illness of childhood, “comparable to other diseases we vaccinate children for.”
The Vaccine Study Center has a 30-year history of enrolling infants, children, teenagers, and adults in vaccine clinical trials, and has been involved in clinical trials for almost every vaccine that has been licensed in the United States. The center is also participating in studies sponsored by the Centers for Disease Control and Prevention to evaluate the vaccines’ real-world effectiveness.
# # #
About the Kaiser Permanente Division of Research
The Kaiser Permanente Division of Research conducts, publishes and disseminates epidemiologic and health services research to improve the health and medical care of Kaiser Permanente members and society at large. It seeks to understand the determinants of illness and well-being, and to improve the quality and cost-effectiveness of health care. Currently, DOR’s 600-plus staff is working on more than 450 epidemiological and health services research projects. For more information, visit divisionofresearch.kaiserpermanente.org or follow us @KPDOR.




This Post Has 2 Comments
I’m so proud of you Dr Yee-Guardino for putting your child first for other parents to feel safe with the vaccine and with our foundation. thank you for all you do Dr Yee-Guardino. 🙂
We are lucky to have tweens like Luci help us confirm the safety and efficacy of life-saving vaccines.
Kudos to all those who participate in vaccine trials!