Kaiser Permanente research leader Tracy Lieu heads high-level effort to move prevention forward
Preventing disease is an essential part of medical care, and research provides the evidence to show which preventive tests and screenings are effective. But major gaps in evidence exist for many common preventive services, including checking vitamin D levels in adults, testing older adults for hearing loss or cognitive impairment, and screening adolescents for unhealthy drug use.
The U.S. Preventive Services Task Force (USPSTF) is the expert group that makes preventive care recommendations for the nation. The task force reviews the evidence for each preventive service and assigns a letter grade recommendation for those with sufficient evidence. For a service with insufficient evidence, the task force issues an “I” statement. For “I” statements that are turned into a letter grade because better evidence has been generated, it takes an average of 8 years, and some preventive services never gain sufficient evidence.

A new National Academies of Sciences, Engineering and Medicine consensus study, headed by Kaiser Permanente Division of Research Director Tracy Lieu, MD, MPH, makes recommendations to accelerate the research needed to fill evidence gaps for clinical preventive services. The National Academies are private, non-profit institutions that provide expert advice to the U.S. to solve problems and inform public policy decisions related to science, technology, and medicine.
Lieu is also the co-lead author of a viewpoint about the findings published January 27 in JAMA. She answered our questions about evidence gaps and the committee’s work.
Can you briefly describe the problem of evidence gaps in clinical prevention?
The U.S. Preventive Services Task Force has a unique role in health care because its statements essentially have the force of law. The Affordable Care Act mandated insurers to cover services the task force recommends. And the task force is quite meticulous about how it relies on evidence to form recommendations. The evidence about a service needs to be quite robust before the task force will determine there is sufficient evidence.
That can lead to challenges for real-life practitioners. If a topic has an insufficient evidence statement, there is more burden on the individual practitioner to decide whether to recommend a test or screening for a given patient.
Our committee was tasked with recommending how to improve the process for producing the research needed to fill evidence gaps, especially gaps that result in insufficient evidence statements. Ultimately, we want there to be sufficient evidence for a definitive recommendation, so we want researchers to focus on the precise set of questions the task force most needs to be addressed.
What were your motivations in getting involved with the committee’s work?
The invitation to chair this committee was a perfect combination of science, policy, and clinical practice, and very similar to the work we do here at the Division of Research. Every day we are thinking about how to create structure that helps researchers and policymakers and clinical practitioners. This National Academies project was something concrete that could help all of these fields collaborate more effectively, and I was pleased to participate.
What was the committee’s process?
Like all National Academies committees, we first heard from the committee sponsors, in this case the National Institutes of Health (NIH) and the Agency for Healthcare Research and Quality (AHRQ), about the tasks we should accomplish. We then held public workshops with invited expert speakers.
So, for instance, we heard from researchers working on colorectal cancer screening, and from the National Cancer Institute officer for that topic. We also gathered information from published and online sources. We created a draft of our major product, a taxonomy — which is a standard list of terms — then assigned ourselves the task of using it. We put the taxonomy through several cycles of testing and refinement before calling it final.
Can you identify a few of its main findings?
Our main product was the taxonomy. It’s like a roadmap that clinical guideline committees, funders, and researchers can use to clearly communicate about the specific areas of research that are needed.
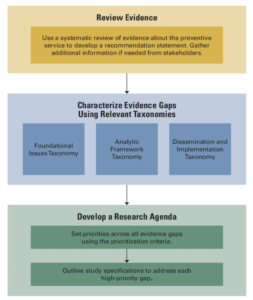 The taxonomy covers three types of research: foundational, analytic framework, and dissemination and implementation. The analytic framework taxonomy matches closely with the way the USPSTF analyzes evidence.
The taxonomy covers three types of research: foundational, analytic framework, and dissemination and implementation. The analytic framework taxonomy matches closely with the way the USPSTF analyzes evidence.
It was significant that we included dissemination and implementation of preventive measures because that sometimes hasn’t been highlighted in research agendas. But real-world studies provide crucial information about the actual impact of a preventive service.
We also recommended that funders such as NIH set aside funding to address high-priority evidence gaps. We suggested they use specific funding mechanisms, such as requests for applications or contracts, to incentivize investigators to conduct the highest priority research studies.
What are you hoping will be done with the recommendations?
Our committee hopes the task force, NIH, AHRQ, and other committees and funders will put the taxonomy into routine use when identifying research needs and priorities. Since the taxonomy offers a shared, consistent set of concepts and language, it can help policymakers, funders, and researchers communicate more effectively and accelerate progress on the needed research.
How might DOR and its investigators be involved in responding to the recommendations?
Our committee suggested more studies of how well USPSTF letter-grade recommendations are implemented in real-world settings. This is an area where Kaiser Permanente researchers excel. For example, in colorectal cancer screening, our research by Doug Corley, T.R. Levin, and others sets an example for the nation. We have outstanding capabilities for high-impact research about both effectiveness and real-life implementation of preventive services.



This Post Has 0 Comments